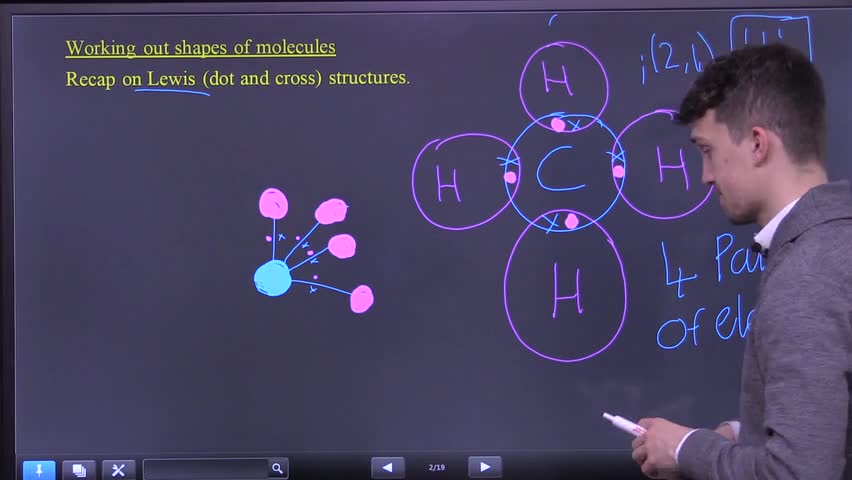
Intro to VSEPR theory and the ball and stick model
📝 My Notes
Auto-saves the current timestamp
Intro to VSEPR theory and the ball and stick model
7:17
About this lesson
Key Moments
No key moments available.
Video 1 of 17
Intro to VSEPR theory and the ball and stick model
7 min
• Selected
Intro to VSEPR theory and electron pair repulsion
4 min
How bonding pairs and lone pairs determine molecular shape
3 min
Linear molecular geometry of beryllium chloride
4 min
How lone pairs affect bond angles in water (H2O)
5 min
Trigonal planar geometry of borane (BH3)
4 min
Trigonal pyramidal shape of NH3 with lone pair repulsion
4 min
Tetrahedral shape of CH4 with four bonding pairs
2 min
Square planar geometry with four bonding pairs
3 min
Trigonal bipyramidal geometry of PCl5 with five bonding pairs
5 min
Octahedral geometry of SF6 with six bonding pairs
3 min
Summary table of molecular geometries and bond angles
3 min
Predicting shapes of Cl2 and AlCl3 using VSEPR theory
5 min
Predicting geometry and bond angles for CH4 and NH3
6 min
Trigonal bipyramidal and octahedral shapes: PCl5 and SF6
6 min
Determining the bent shape and bond angle of water (H2O)
3 min
Determining the T-shape geometry of ClF3 using VSEPR
19 min