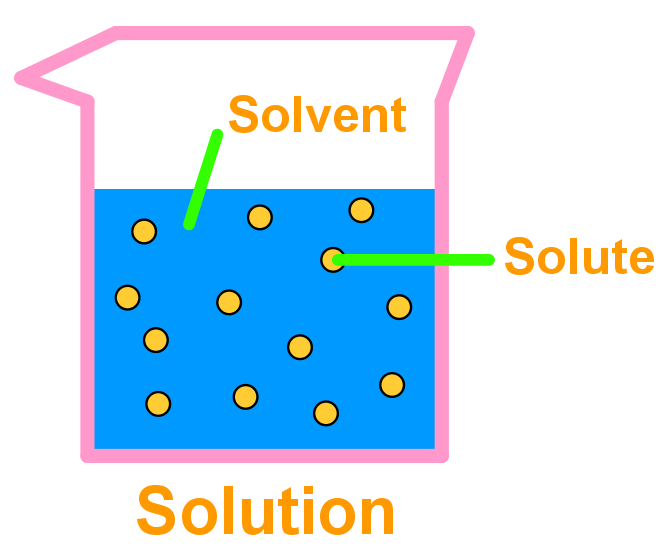
Introduction to solution chemistry and solubility
All You Need in One PlaceEverything you need for Year 6 maths and science through to Year 13 and beyond. | Learn with ConfidenceWe’ve mastered the national curriculum to help you secure merit and excellence marks. | Unlimited HelpThe best tips, tricks, walkthroughs, and practice questions available. |
Make math click 🤔 and get better grades! 💯Join for Free
0/4
Intros
0/5
Examples
Lessons
- Recall the key definitions of solution chemistry.
- Recall how chemists measure solubility of chemical substances.
What factors need to be mentioned when chemists quote or report a chemical's solubility? - Recall the difference between a saturated and unsaturated solution.
A student has two glasses of a salt solution, A and B, completely dissolved in water. One of them is saturated and the other is unsaturated.