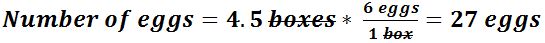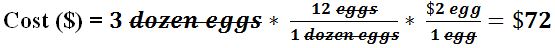Unit conversions in chemistry
All in One PlaceEverything you need for better grades in university, high school and elementary. | Learn with EaseMade in Canada with help for all provincial curriculums, so you can study in confidence. | Instant and Unlimited HelpGet the best tips, walkthroughs, and practice questions. |
Make math click 🤔 and get better grades! 💯Join for Free
0/3
Intros
0/10
Examples
Lessons
- Apply the conversion factor method to simple calculations.
Use the unit conversion method to answer the following problems.- If a car can travel 75 kilometres in 1 hour, how far can it travel in 4.5 hours?
- An electronics store has an offer that sells two TVs for $430. How much would 8 TVs cost using this deal?
- At a market, a man traded 8 apples for 14 oranges. How many oranges can he get if he traded 22 apples at this rate?
- Apply the conversion factor method to chemistry-related calculations.
Use the unit conversion method to answer the following problems.- If a container holds 3.5 dozen oxygen atoms, how many oxygen atoms in total will 3 full containers have in them?
- If one mole of oxygen gas has a mass of 32 g, what is the mass of 3.5 moles of oxygen gas?
- If one molecule of white phosphorus has 4 atoms of phosphorus in it, how many molecules of white phosphorus would be needed to have 72 atoms of phosphorus?
- If one mole of hydrogen gas fills up 22.4 L in a gas canister, how many moles of hydrogen gas would fill up a gas canister 180 L in size?
- Apply the conversion factor method to chemistry-related calculations with SI units.
Use the unit conversion method to answer the following problems.- 4.7 moles of carbon dioxide gas has a mass of 206.8 g. What is the mass of 1 mole of carbon dioxide gas?
- Gold has a density of 19.3 grams per millilitre (g/mL), what would the volume be of 62 grams of gold?
- If an acid has a concentration of 3 moles per litre, how many litres of acid would I need to have 1.8 moles of acid?